Department of Pharmacology and Toxicology, Medical College of Wisconsin, Milwaukee, WI, USA
chillard@mcw.edu

Introduction
One of the environmental factors that precipitates and exacerbates mental illnesses, including depression, anxiety disorders, and substance abuse, is repeated life stress. For example, homotypic stressors that occur on a daily basis, such as poverty or medical problems, are associated with increased depressive symptoms [1]. Not everyone exposed to stress has pathological consequences. The factors that protect against the "allostatic" load of repeated stress include: an ability to habituate to the stress, maintenance of hedonic tone and reward in the face of stress, and extinguishing of fearful memories [2]. It is clear from preclinical studies that CB1 receptor signalling increases sensitivity to reward [3] and is critical for the extinction of aversive memories [4]. Similarly, data is accumulating to support a role for the endocannabinoid system (ECS) in reducing stress responsivity, including a decrease in endocrine and behavioural responses to the initial presentation of the stress and in the development of habituation.
Key Points
- Stressful life events or situations contribute to many human diseases, including depression, anxiety, and cardiovascular disease.
- Data from animal studies suggests that the endogenous cannabinoid signalling system is a mechanism by which stress is buffered or dampened.
- In mice exposed to an acute stress, loss of endocannabinoid signalling, either through pharmacological blockade or genetic deletion, results in an exaggerated activation of the HPA axis, as well as exaggerated behavioural responses evoked by the stress or threat.
- Repeated exposure to the same stress results in habituation. Pharmacological blockade of endocannabinoid signalling reinstates the behavioural response to stress in habituated mice. In other words, we hypothesize that habituation is accompanied by an activation of endocannabinoid signalling in response to the stress, which serves to reduce the response.
- Repeated exposure to variable stressors and administration of the glucocorticoid corticosterone results in decreased CB1 receptor expression in the hippocampus and an increase in the prefrontal cortex. This data is consistent with the behavioural effects of chronic stress on inducing symptoms of depression.
State of the Art
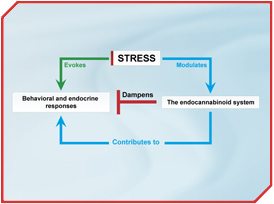
A simple model that reflects our current understanding about the relationships between stress and the ECS is presented in the Figure.
The interactions between the ECS and stress are bidirectional; stress alters the ECS and the ECS alters stress responses. As a result of this interrelationship, the ECS is in an excellent position to provide feedback to the stress circuit. This speculation has been confirmed by many types of studies.
In acute stress, a primary role of the ECS is to dampen hypothalamic-pituitary-adrenal (HPA) axis activation. Inhibition of the ECS increases and augmentation of the ECS decreases HPA axis activation [5, 6]. Acute stress also produces behavioural effects, including reduced exploration, defensive postures, and anhedonia. These behavioural manifestations of stress are also dampened by the ECS [7-9]. On the other hand, data demonstrating that the ECS plays a role in dampening immobility responses in the forced swim assay (interpreted as a measure of despair in response to a stressful situation) is not consistent. Some investigators find that CB1 activation reduces immobility [10, 11] while others report that CB1 receptor inhibition reduces immobility [12]. These discrepancies likely lie in the lack of reliability of immobility as a measure of stress in rodents. In summary, the available data indicates that the ECS dampens stress responses.
 [Click to enlarge]
[Click to enlarge]
Tissue contents of anandamide (AEA) are reduced in the hippocampus and amygdala and 2-arachidonoylglycerol (2-AG) is reduced in the hypothalamus following acute stress exposure [13]. On the one hand, this data is puzzling because the hormonal and behavioural data described above suggest that ECS signalling becomes activated during stress. However, an alternative explanation is that ECS signalling is high under non-stressed conditions and that the effects of stress are related to the degree to which endocannabinoid presence at the receptor is decreased [6].
Chronic exposure to the same stressor produces habituation, revealed as a reduction in the hormonal and behavioural consequences of stress exposure. Data is accumulating that habituation requires the ECS. For example, habituation to neuronal activation in the prefrontal cortex is blocked by CB1 receptor antagonist treatment [14]. Furthermore, preliminary data from our laboratory demonstrates that habituation to HPA axis activation by restraint is lost in CB1 receptor null mice. Repeated restraint stress alters tissue contents of the endocannabinoids in a region and ligand-dependent manner [14, 15]. Interestingly, the changes are consistent with the hypothesis that repeated exposure to the same stressor increases activation of the ECS, due to enhanced ligand release, which down-regulates stress responses.
Chronic exposure of rodents to variable stressors at random times (CUS) results in behavioural changes that mimic those seem in human depression. CUS, which results in a chronic elevation of serum corticosterone, has significant effects on the ECS. CB1 receptor density is decreased in the hippocampus [16] and increased in the prefrontal cortex [17] in rats exposed to CUS. We have also identified similar changes in mice at the mRNA level. The effect of CUS on hippocampal CB1 receptor density is mimicked by chronic treatment with corticosterone [18]. CUS also causes perservatory behaviour in the Morris water maze test, which is reduced when a CB1 agonist is present. This data suggests that down-regulation of CB1 signalling, perhaps in the hippocampus, contributes to some of the behavioural manifestations of chronic stress. Repeated restraint stress under conditions in which there was no habituation results in decreased sucrose consumption in mice. This effect is reversed by CB1 receptor activation [9], providing another example of a role for dysregulation of the ECS in the consequences of stress.
Priority for Future Studies
Our understanding of the interactions between stress and the ECS are at the beginning of their maturity. Better tools are needed to apply to this issue. For example, selective and efficacious pharmacological inhibitors of the synthesis and degradation of the endocannabinoid ligands are a high priority for many in vivo studies. In addition, more selective genetic deletions will be helpful in sorting out the neurocircuitry of the interactions of the ECS with stress. Most importantly, we need to understand whether the role of the ECS as a stress buffer contributes to human disease. For example, does early life stress alter the ECS and does this contribute to an increased propensity for depression and addiction? Can augmentation of the ECS be used as a therapeutic approach for the treatment of stress-related diseases, including depression and anxiety?
References
- Caspi A, Sugden K, Moffitt TE, et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 2003; 301: 386-9.
- Charney DS. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am J Psychiatry 2004; 161: 195-216.
- Gardner EL. Endocannabinoid signaling system and brain reward: emphasis on dopamine. Pharmacol Biochem Behav 2005; 81: 263-84.
- Marsicano G, Wotjak CT, Azad SC, et al. The endogenous cannabinoid system controls extinction of aversive memories. Nature 2002; 418: 530-4.
- Barna I, Zelena D, Arszovszki AC, et al. The role of endogenous cannabinoids in the hypothalamo-pituitary-adrenal axis regulation: in vivo and in vitro studies in CB1 receptor knockout mice. Life Sci 2004; 75: 2959-70.
- Patel S, Roelke CT, Rademacher DJ, et al. Endocannabinoid signaling negatively modulates stress-induced activation of the hypothalamic-pituitary-adrenal axis. Endocrinology 2004; 145: 5431-8.
- Hill MN, Kambo JS, Sun JC, et al. Endocannabinoids modulate stress-induced suppression of hippocampal cell proliferation and activation of defensive behaviours. Eur J Neurosci 2006; 24: 1845-9.
- Patel S and Hillard CJ. Pharmacological evaluation of cannabinoid receptor ligands in a mouse model of anxiety: further evidence for an anxiolytic role for endogenous cannabinoid signaling. J Pharmacol Exp Ther 2006; 318: 304-11.
- Rademacher DJ and Hillard CJ. Interactions between endocannabinoids and stress-induced decreased sensitivity to natural reward. Prog Neuropsychopharmacol Biol Psychiatry 2007; 31: 633-41.
- Gobbi G, Bambico FR, Mangieri R, et al. Antidepressant-like activity and modulation of brain monoaminergic transmission by blockade of anandamide hydrolysis. Proc Natl Acad Sci U S A 2005; 102: 18620-5.
- Hill MN and Gorzalka BB. Pharmacological enhancement of cannabinoid CB1 receptor activity elicits an antidepressant-like response in the rat forced swim test. Eur Neuropsychopharmacol 2005; 15: 593-9.
- Shearman LP, Rosko KM, Fleischer R, et al. Antidepressant-like and anorectic effects of the cannabinoid CB1 receptor inverse agonist AM251 in mice. Behav Pharmacol 2003; 14: 573-82.
- Gorzalka BB, Hill MN and Hillard CJ. Regulation of endocannabinoid signaling by stress: implications for stress-related affective disorders. Neurosci Biobehav Rev 2008; 32: 1152-60.
- Patel S, Roelke CT, Rademacher DJ, et al. Inhibition of restraint stress-induced neural and behavioural activation by endogenous cannabinoid signalling. Eur J Neurosci 2005; 21: 1057-69.
- Rademacher DJ, Meier SE, Shi L, et al. Effects of acute and repeated restraint stress on endocannabinoid content in the amygdala, ventral striatum, and medial prefrontal cortex in mice. Neuropharmacology 2008; 54: 108-16.
- Hill MN, Patel S, Carrier EJ, et al. Downregulation of endocannabinoid signaling in the hippocampus following chronic unpredictable stress. Neuropsychopharmacology 2005; 30: 508-15.
- Bortolato M, Mangieri RA, Fu J, et al. Antidepressant-like activity of the fatty acid amide hydrolase inhibitor URB597 in a rat model of chronic mild stress. Biol Psychiatry 2007; 62: 1103-10.
- Hill MN, Carrier EJ, Ho WS, et al. Prolonged glucocorticoid treatment decreases cannabinoid CB1 receptor density in the hippocampus. Hippocampus 2008; 18: 221-6.



